These coordinates are similar to the ones used in GPS devices and most smart phones that track positions on our (nearly) spherical earth, with the two angular coordinates specified by the latitude and longitude, and the linear coordinate specified by sea-level elevation. Central potentials have spherical symmetry, and so rather than specifying the position of the electron in the usual Cartesian coordinates ( x, y, z), it is more convenient to use polar spherical coordinates centered at the nucleus, consisting of a linear coordinate r and two angular coordinates, usually specified by the Greek letters theta (θ) and phi ( Φ). Potentials of the form V( r) that depend only on the radial distance r are known as central potentials. Because the electrostatic potential has the same form as the gravitational potential, according to classical mechanics, the equations of motion should be similar, with the electron moving around the nucleus in circular or elliptical orbits (hence the label “planetary” model of the atom). The electrostatic potential is also called the Coulomb potential. Since forces can be derived from potentials, it is convenient to work with potentials instead, since they are forms of energy. The electrostatic force has the same form as the gravitational force between two mass particles except that the electrostatic force depends on the magnitudes of the charges on the particles (+1 for the proton and −1 for the electron) instead of the magnitudes of the particle masses that govern the gravitational force. 
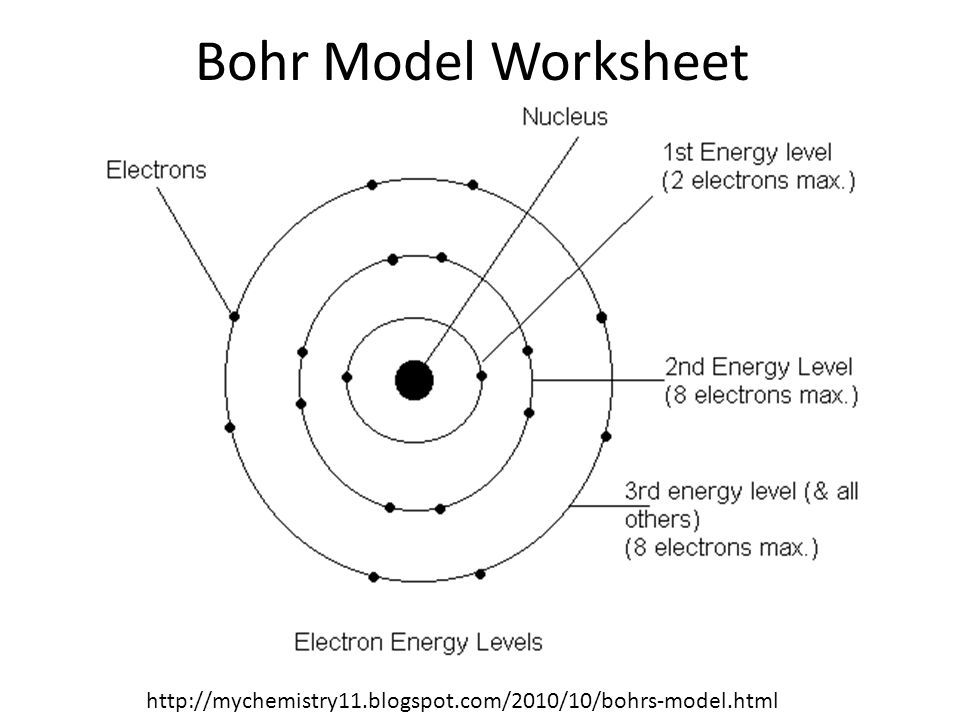
The electrostatic force attracting the electron to the proton depends only on the distance between the two particles. The simplest atom is hydrogen, consisting of a single proton as the nucleus about which a single electron moves. This picture was called the planetary model, since it pictured the atom as a miniature “solar system” with the electrons orbiting the nucleus like planets orbiting the sun. Use the Rydberg equation to calculate energies of light emitted or absorbed by hydrogen atomsįollowing the work of Ernest Rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established.Describe the Bohr model of the hydrogen atom.Since oxygen has got 6 electrons remaining, these electrons occupy the valence (outermost)shell of the of the oxygen atom, and the radius these electrons describe, define the atomic radius of ATOMIC oxygen.Įlectronic interactions between different atoms can form chemical bonds.for oxygen the most stable electronic arrangement occurs when a pair of such atoms make music to form a dioxygen molecule, #O_2#.By the end of this section, you will be able to: Two electrons can fit into the first level, BUT up to 8 electrons can fit in the next available level(s). modern views of electronic structure hold that the electrons are confined to various energy levels. Around the nuclear core, 8 electrons, 8 fundamental negatively charged particles of negligible mass are conceived to whizz about. Interactions between protons, and neutrons, AT EXCEPTIONALLY short nuclear ranges, are strong enough to overcome the electrostatic repulsion between electrically charged particles of the same charge. So what does #Z# represent? It is the atomic number, the number of massive, positively charged particles that comprise the oxygen nucleus.these particles, these nuclear protons are packed into a dense nuclear core.The most common isotope of oxygen also packs 8 neutrons, 8 massive neutrally charged particles into the core, i.e.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
